Overview
Epigenetic regulatory mechanisms represent attractive targets for new therapeutic approaches to a variety of human health problems due to the global nature of gene expression modification achieved through their use. Histone deacetylase (HDAC) inhibitors (HDACi) have been approved for use in the treatment of certain T-cell lymphomas, but class I HDACi have been shown to do the following:
- Upregulate fetal hemoglobin expression
- Synergize with immuno-oncology approaches
- Trigger both pro- and anti-inflammatory reactions
- Activate latent pools of HIV
- Interfere with Transcriptional Dysregulation
Also, currently approved HDACi have significant toxicity, which is not acceptable for the treatment of chronic, nonlethal disease. Cetya’s HDAC inhibitor portfolio includes 30+ compounds and provides a systematic approach to developing class I HDACi with an acceptable toxicity profile to treat chronic disorders.
Indications
Hemoglobinopathies
Hemoglobinopathies such as sickle cell disease and b-thalassemias are driven by a mutation in the gene for adult hemoglobin, although the fetal hemoglobin in these patients is normal and functional. In addition to having a reduced ability to bind oxygen, the mutated form of the b-globin protein is subject to polymerization when not transporting oxygen. These polymers precipitate, resulting the sickled shape of the red cells and the increased rigidity of the sickled red cell membrane. The sickled red cells get stuck in capillary beds, clogging the vessel and severely restricting blood flow downstream of the clog. This leads to oxygen deficit, cell death and tissue and organ damage. These events are termed vaso-occlusive crises (VOC) and are excruciatingly painful for the patient. The tissue and organ damage is progressive, accumulating over time and ultimately resulting in organ failure.
Fetal hemoglobin has been shown to interfere in the polymerization process, reducing the extent of clogging and thus mitigating the symptoms of sickle cell disease. A study published over 25 years ago found that patients with lower levels of fetal hemoglobin survived for an average of 40 years whereas patients with a higher level of fetal hemoglobin lived for 55-60 years.1 CT-101 has been found to elevate the level of fetal hemoglobin in both erythroid progenitor cells take from sickle cell or thalassemia patients and also in mouse models of SCD.
The Class I HDACs 1, 2 and 3 are present in the repressor complex that sits on the gene promoter for fetal hemoglobin, and studies have shown that exposure of these complexes to a Class I HDAC inhibitor causes the complex to dissociate from the promoter allowing gene expression to reinitiate. Cetya has data showing that CT-101 can increase fetal hemoglobin expression two- to three-fold without appreciable bone marrow toxicity. The lack of toxicity is an important consideration for any drug therapy that requires chronic administration such as that anticipated for sickle cell disease.

Top panel: Fetal globin (γ-globin) expression is repressed at birth, and adult globin (β-globin) expression facilitates hemoglobin production. Fetal globin expression is repressed via binding of the direct repeat erythroid-definitive (DRED) complex to the direct repeat (DR) elements in the γ-globin promoter. HDACs alone or in complex act as corepressors to block fetal globin expression. Middle panel: Hemoglobinopathies occur when β-globin expression is low (β-thalassemia) or when a mutated form of β-globin is produced (sickle cell disease). Bottom panel: HDAC inhibitors have been shown to block repression of γ-globin, resulting in expression of fetal globulin, which reduces symptoms of hemoglobinopathies. Figure derived from Suzuki et al.2
References:
- Platt, O.; Brambilla, D.; Rosse, W.: et al., Mortality in Sickle Cell Disease – Life Expectancy and Risk Factors for Early Death. N Engl J Med, 1994 330: 1639-1644.
- Suzuki, M.; Yamamoto, M.; and Engel, J. D., Fetal Globin Gene Repressors as Drug Targets for Molecular Therapies To Treat the β-Globinopathies. Molecular and Cellular Biology, 2014, 34(19): 3560-3569. >>
Immuno-oncology
HDAC inhibitors have the potential to upregulate MHC Class I expression in tumor cell membranes. This has been observed with Cetya’s compound CT-102 by one of our collaborators. Higher levels of MHC Class I in the tumor cell membrane will make the tumor more visible to the host immune system, thereby potentiating the effects of CAR T cell therapies or other immuno-oncology approaches.
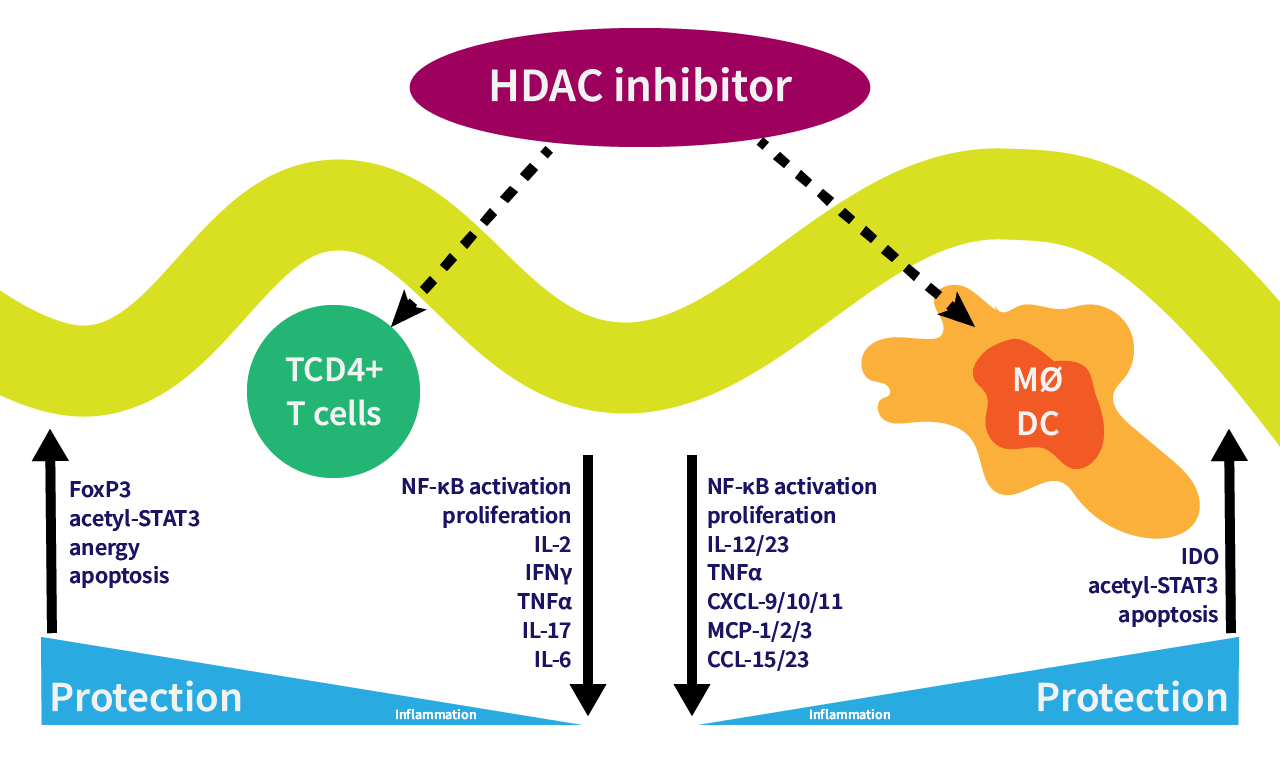
Chronic Inflammation
Chronic inflammatory diseases such as rheumatoid arthritis are characterized by periodic episodes of inflammation driven in part by the systematic release of cytokines (i.e., cytokine storms). Central to this process is the macrophage, a phagocytic white blood cell that is part of the innate immune response. Resting macrophages are activated following exposure to either TNF-α or interferon-γ. These activated macrophages then react to certain stimuli, such as endotoxin, by releasing various cytokines that drive the cytokine storm that results in local inflammation.
Class I histone deacetylase (HDAC) inhibitors (HDACi) have been shown to have both pro- and anti-inflammatory activities. Cetya’s HDACi portfolio of 30+ compounds allows a systematic approach to developing class I HDACi that maximize anti-inflammatory activity. The largazole-based scaffold provides a building block for a directed structure-activity relationship (SAR) lead optimization program. The development program will be based on the specific biological readouts of macrophage activation and cytokine secretion. In addition, currently approved HDAC inhibitors such as vorinostat and romidepsin have significant toxicities that are unacceptable for treatment of a chronic, nonlethal disease.
The macrocyclic ring within the largazole structure can be modified in multiple places to incorporate a targeting moiety, allowing the resulting derivative to target a cell type of interest; in this case, use of a high-mannose group to access resting macrophages through the mannose receptor. Targeting is the classic approach to reduce toxicity, the driver of monoclonal antibody therapy as well as alternate formulation strategies such as liposomes or nanoparticles.
This systematic approach provides the opportunity to utilize HDACi for the treatment of chronic disorders where, to date, their use has not been feasible due to systemic toxicities.
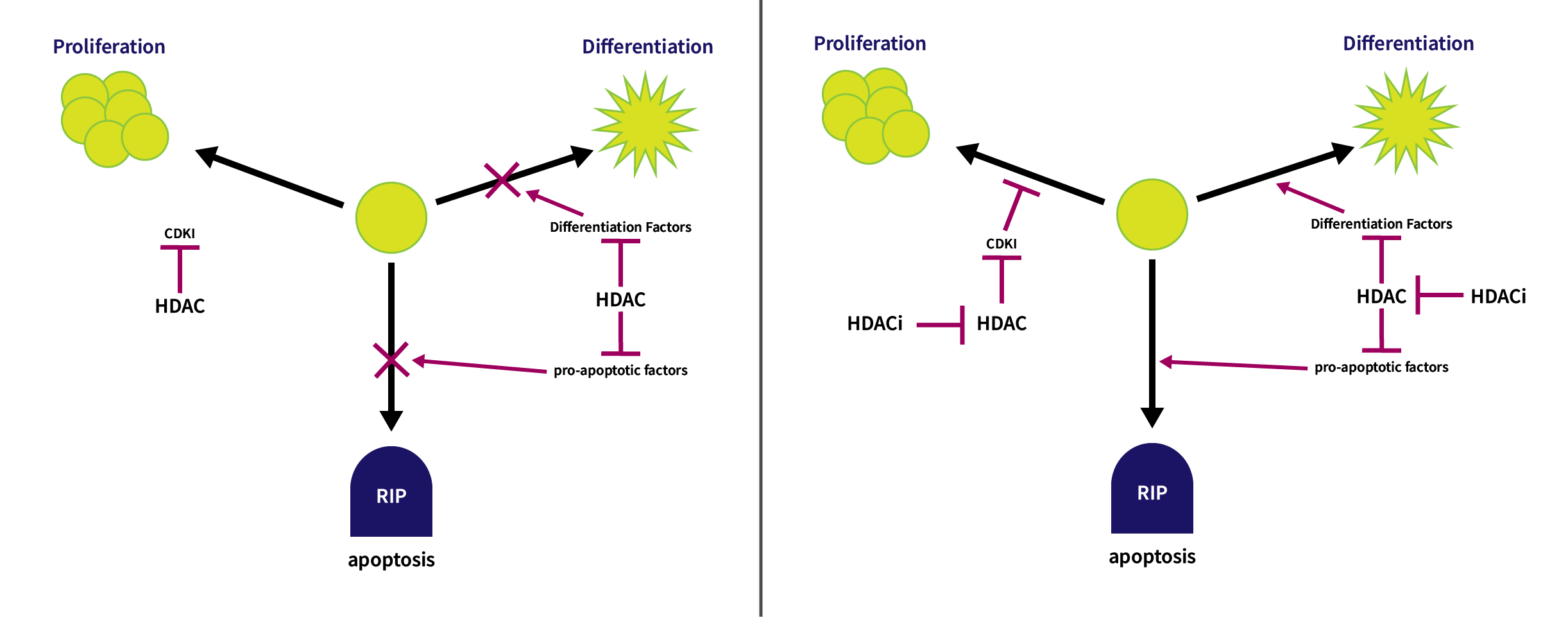
Oncology
Cancer remains a major unmet medical need, both in the US and worldwide. The American Cancer Society estimates that over 1.7 million people in the US will be diagnosed with cancer in 2018, and roughly 610,000 deaths from cancer will occur1; one in five people in the US will die of cancer2.
Understanding the molecular basis of cancer has increased dramatically in the last decade with the availability of the complete sequence of the human genome and the significant decrease in the cost of obtaining human DNA sequence information. This understanding has driven the desire for the development of “personalized medicine”, therapeutic agents tailored to the molecular defects found in an individual’s tumor. As a result, virtually all of the major pharmaceutical and biotech companies worldwide have identified oncology as an area of major interest for the development of new therapeutics.
The so-called liquid tumors, or tumors of the blood and lymphatic systems, including leukemia, lymphomas, and multiple myeloma, are cancer types where histone deacetylase (HDAC) inhibitors (HDACi) have shown clinical benefit and gained FDA approval. This collection of tumor indications represents roughly 10% of all cancer incidence and mortality.
HDACs act as one level of control on the pattern of gene expression in all types of human cells. Cancer cells have been shown to downregulate the expression level of proteins that interfere with their ability to survive and to upregulate the expression level of proteins that enhance their ability to survive. This regulation of gene expression has been linked in part to HDACs, hence the interest in HDACi as agents to potentially reverse these defense mechanisms.
HIV Therapy
More than 35 million people have died from AIDS, and there is no known cure for the disease despite over 30 different anti-HIV drugs approved for human use1. The current clinical strategy is to treat patients with a combination of commercial drugs that target different points in the viral life cycle. This antiretroviral therapy (ART) strategy was introduced in the 1990s and has resulted in a dramatic reduction in HIV-associated mortality and morbidity2. ART targets different points in the viral life cycle, including:
- Cell fusion
- Reverse transcriptase of viral RNA
- Integration of viral DNA
- Inhibition of viral processing (via protease inhibitors)
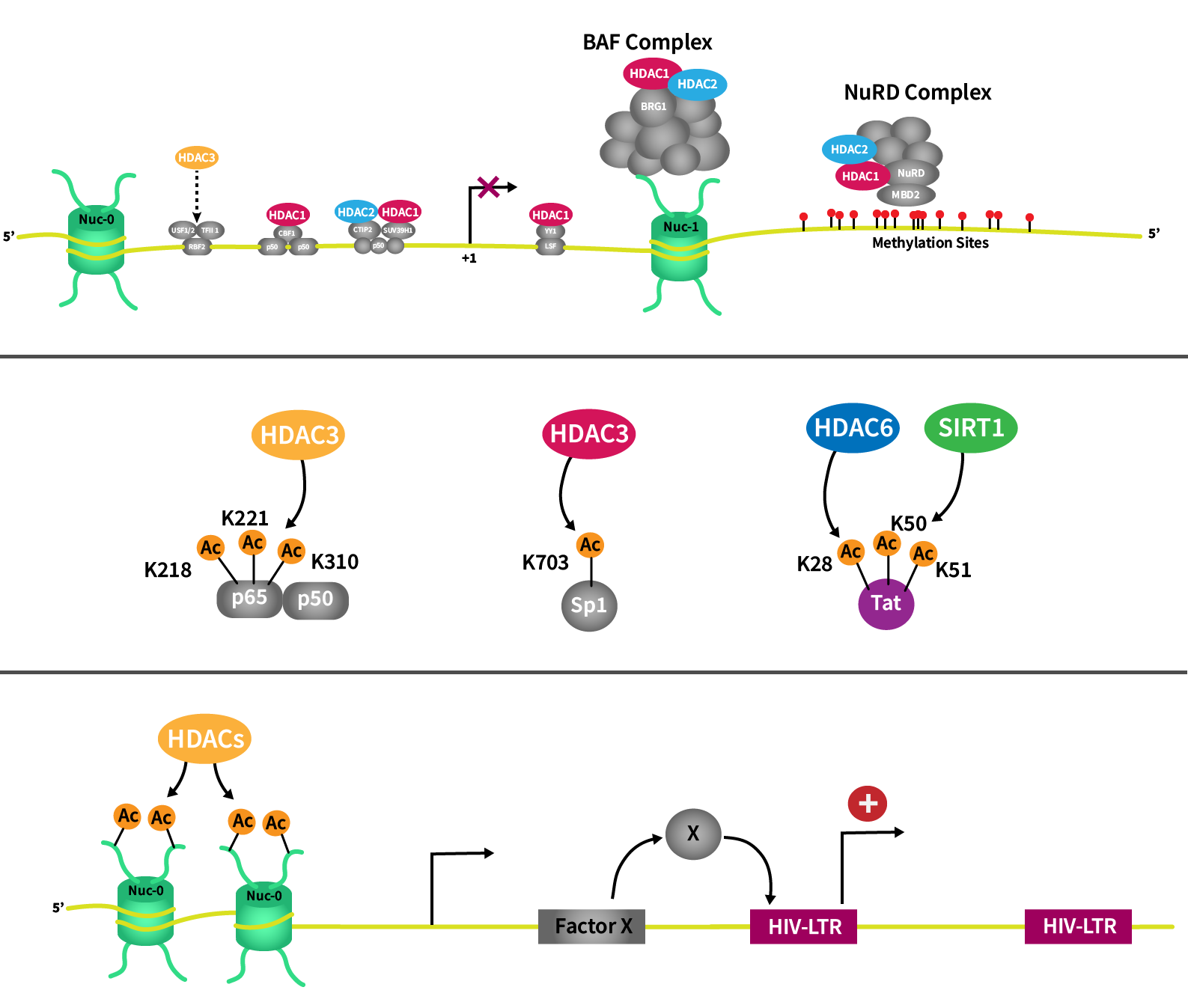
Latent pools of the virus are generally thought to be established in cells such as resting CD4+ memory T cells during the initial acute phase of HIV viral infection. These cell pools are resistant to ART and are transcriptionally silent until externally stimulated3. Transcriptional silencing in latent cell pools is maintained at the HIV long terminal repeat (LTR) promoter, where nucleosome-bound conformations prevent transcription and maintain the silent integrated viral DNA in a resting state. The latent resting pools represent an extremely stable reservoir with a half-life of 4 years or longer1.
There is mounting scientific evidence that histone deacetylases (HDACs) are actively involved in maintaining the transcriptionally silent cell pools. During the acute phase of HIV infection, the latent cell pools are maintained in a quiescent state or activated via complex interactions of the nucleosome, HIV regulatory proteins and eukaryotic transcription factors. The action of class 1 HDACs is required to maintain pro-viral quiescence, and studies have demonstrated that HDAC inhibition results in LTR activation and transcription of the latent pro-viral DNA1.
Several HDAC inhibitors (HDACi) have been evaluated for their ability to reactivate transcriptionally silent reservoirs in cells and HIV-infected patients2. These agents include vorinostat, givinostat, panobinostat, entinostat, mocetinostat, and romidepsin2. Use of first-line HDACi to activate latent viral pools may be hampered by toxicity issues associated with using these compounds at a clinically effective dose.
Together, these data demonstrate the validity of using HDACi to activate latent pro-viral HIV pools in HIV-infected patients. Cetya’s compounds exhibit enhanced class I specificity, best-in-class potency and reduced toxicity profiles that might be ideal candidates to use in combination with ART strategies to eradicate latent HIV viral pools and and potentially lead to a cure for HIV.
References:
- Ruelas, D.; and W. Greene, An Integrated Overview of HIV-1 Latency. Cell, 2013, 155:519-529. >>
- Shirakawa, K.: Chavez, L.; Hakre, S.; Calvanese, V.; and E. Verdin, Reactivation of latent HIV by histone deacetylase inhibitors. Trends Microbiol. 2013, 21:277-285. >>
- Archin, N.; Espeseth, A.; Parker, D.; Cheema, M.; Hazuda, D.; and D. M. Margolis, Expression of Latent HIV Induced by the Potent HDAC Inhibitor Suberoylanilide Hydroxamic Acid. Aids Res. Human Retroviruses 2009, 25:207-212. >>
Neurodegenerative Disease
There is growing evidence that histone deacetylace (HDAC) inhibitors (HDACi) might be effective in treating certain neurodegenerative diseases. The current available treatments for Alzheimer’s, Parkinson’s, amyloid lateral sclerosis (ALS), and Huntington’s diseases do not cure the ailments but rather inhibit progression of the patient’s disease1. Scientific evidence continues to support the hypothesis that HDAC dysregulation plays a central role in the development and progression of neurodegenerative disorders and the resulting clinical pathologies.
Current evidence suggests that HDACi are potentially relevant clinical candidates to treat neurodegenerative diseases including Huntington’s2, Niemann-Pick Type C3-4 and Amyotrophic Lateral Sclerosis5.
References:
- F. Coppede, The potential of epigenetic therapies in neurodegenerative diseases. Frontiers in Genetics, 2014, 5:1-8. >>
- Sharma, S.; and R. Taliyan, Transcriptional Dysregulation in Huntington’s Disease: The Role of Histone deacetylases. Pharmacol. Res, 2015, 100:157-169. >>
- Helquist, P.; Maxfield, F. R.; Wiech, N. L.; and O. Wiest, Treatment of Niemann-Pick Type C Disease by Histone Deacetylase Inhibitors. Neurotherapeutics, 2013, 10:688-697. >>
- Wehrmann, Z. T.; Hulett, T. W.; Huegel, K. L.; Vaughan, K. L.; Wiest, O.; Helquist, P.; and H. Goodson, Quantitative Comparison of the Efficacy of Various Compounds in Lowering Intracellular Cholesterol Levels in Niemann-Pick Type C Fibroblasts. PLOS One, 2012, 7:1-9. >>
- Lee, J.; Ryu, H.; Keum, G.; Yoon, Y. J.; Kowall, N. W.; and H. Ryu, Therapeutic Targeting of Epigenetic Components in Amyotrophic Lateral Sclerosis (ALS). Curr. Med. Chem., 2014, 21:3576-3582. >>
